Direct tracking of reverse-transcriptase speed and template sensitivity: implications for sequencing and analysis of long RNA molecules
Li-Tao Guo, Sara Olson, Shivali Patel, Brenton R Graveley and Anna Marie Pyle
Nucleic Acids Res 2022 (50) : 6980-6989 |
|
A minimal RNA ligand for potent RIG-I activation in living mice
Melissa M. Linehan, Thayne H. Dickey, Emanuela S. Molinari, Megan E. Fitzgerald, Olga Potapova, Akiko Iwasaki and Anna Marie Pyle
Sci Adv. 2018 (4) : e1701854 |
|
Environmentally triggerable retinoic acid-inducible gene I agonists using synthetic polymer overhangs
Christian R Palmer, Max E Jacobson, Olga Fedorova, Anna Marie Pyle and John T. Wilson
Bioconjug. Chem. 2018 (29) : 742-747 |
|
Selective RNA targeting and regulated signaling by RIG-I is controlled by coordination of RNA and ATP binding
Megan E. Fitzgerald, David C. Rawling, Olga Potapova, Xiaoming Ren and Anna Marie Pyle
Nucleic Acids Res. 2017 (45) : 1442-1454 |
|
Establishing the role of ATP for the function of the RIG-I innate immune sensor
David Rawling, Megan Fitzgerald and Anna Marie Pyle
Elife 2015 (4) : E09391 |
|
The RIG-I ATPase core has evolved a functional requirement for alosteric stabilization by the pincer domain
David Rawling, Andrew Kohlway, Dahai Luo, Steve C. Ding and Anna Marie Pyle
Nucleic Acids Res. 2014 (42) : 11601-11611 |
|
Parts, assembly and operation of the RIG-I family of motors
David C. Rawling and Anna Marie Pyle
Curr Opin Struct Biol. 2014 (25) : 25-33 |
|
Dicer-related helicase 3 (DRH-3) forms an obligate dimer for recognizing 22G-RNA
Megan Fitzgerald, Adriana Vela and Anna Marie Pyle
Nucleic Acids Res. 2014 (42) : 19-30 |
|
Defining the functional determinants for RNA surveillance by RIG-I
Andrew Kohlway, Dahai Luo, David C. Rawling, Steve C. Ding and Anna Marie Pyle
EMBO Rep. 2013 (14) : 772-779 |
|
Duplex RNA activated ATPases (DRAs): Platforms for RNA sensing signaling and processing
Dahai Luo, Andrew Kohlway and Anna Marie Pyle
RNA Biol. 2013 (10) : 111-120 |
|
The thermodynamic basis for viral RNA detection by the RIG-I innate immune sensor
Adriana Vela, Olga Fedorova, Steve O. Ding and Anna Marie Pyle
J Biol Chem. 2012 (287) : 42564-42573 |
|
Visualizing the determinants of viral RNA recognition by innate immune sensor RIG-I
Dahai Luo, Andrew Kohlway, Adriana Vela and Anna Marie Pyle
Structure 2012 (20) : 1983-1988 |
|
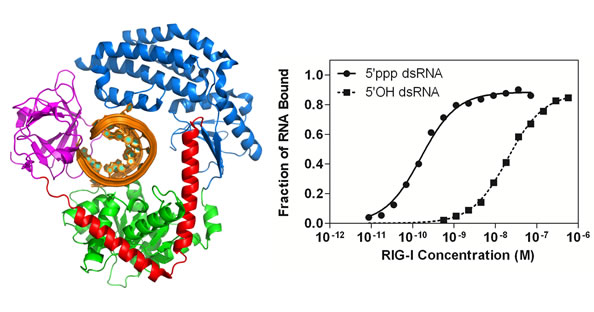
Structural insights into RNA recognition by RIG-I
Dahai Luo, Steve C. Ding, Adriana Vela, Andrew Kohlway, Brett D. Lindenbach and Anna Marie Pyle
Cell 2011 (147) : 409-422 |
|
RNA helicases and remodeling proteins
Anna Marie Pyle
Curr Opin Chem Biol. 2011 (15) : 636-642 |
|